 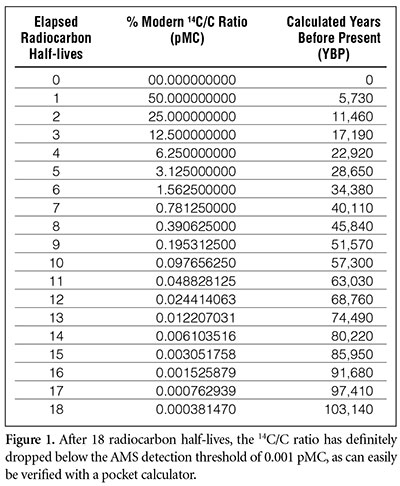
Examination of tissue samples performed independently at Zurich and Oxford revealed that 47% of the C-14 present in the body at the time of his death had decayed. 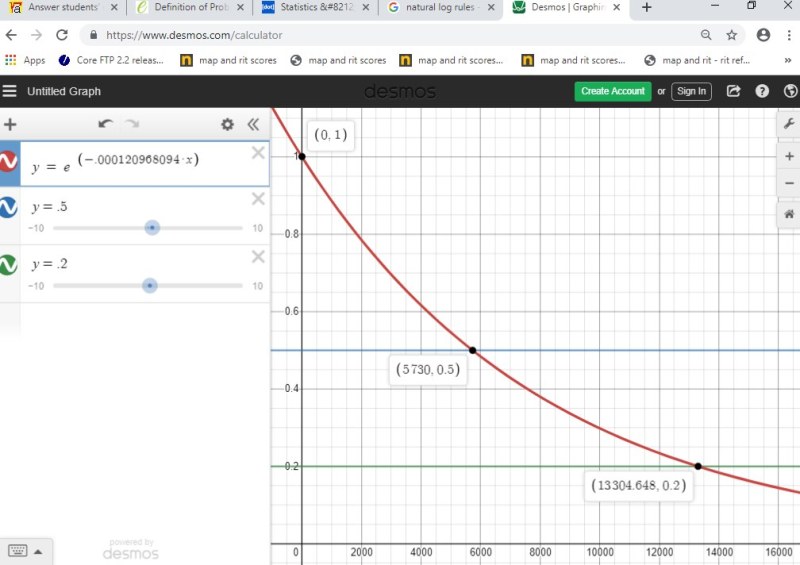
It decays into nitrogen-14 ( 14 N) through beta decay. A well-known historian in Innsbruck, Austria, determined that the man had lived in the Bronze Age, which started about 2000 B.C. The isotopes carbon-12 and carbon-13 are stable, but carbon-14 is radioactive. While the half-life of Anavar is between 9 and 14 hours, meaning it leaves. In 1991, the body of a man was found in melting snow in the Alps of Northern Italy. Here are some tips fThe formulas of algebra are used every day in real life. Determine the half-life of(that is, the time it takes for half of the C-14 to decay). We are told that x(t) satisfies the differential equation Let x(t) be the proportion of the original C-14 still present t years after death. Therefore, carbon dating enables us to calculate the time at which an organism died. After the death of an organism, exchange stops, and the carbon decays. This radiocarbon arises from cosmic-ray bombardment in the upper atmosphere and enters living systems by exchange processes. Question:The carbon in living matter contains a minute proportion of the radioactive isotope C-14. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
